Biopharmaceutical Analytical Testing Services Market Size 2026-2030
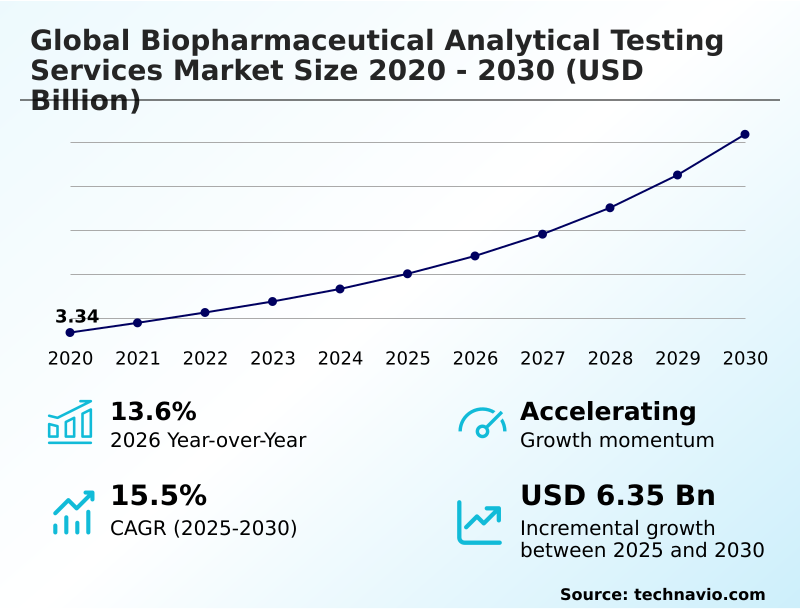
The Biopharmaceutical Analytical Testing Services Market size was valued at USD 6.01 billion in 2025, growing at a CAGR of 15.5% during the forecast period 2026-2030.
Major Market Trends & Insights
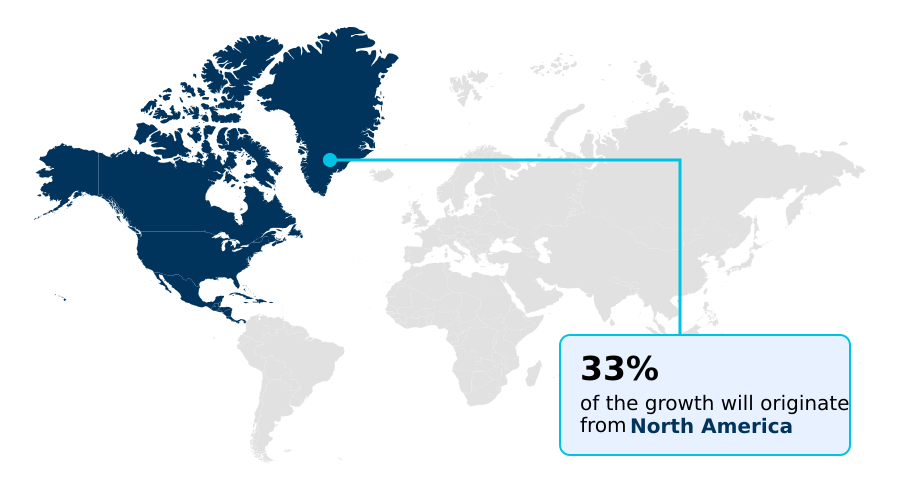
- North America dominated the market and accounted for a 33.3% growth during the forecast period.
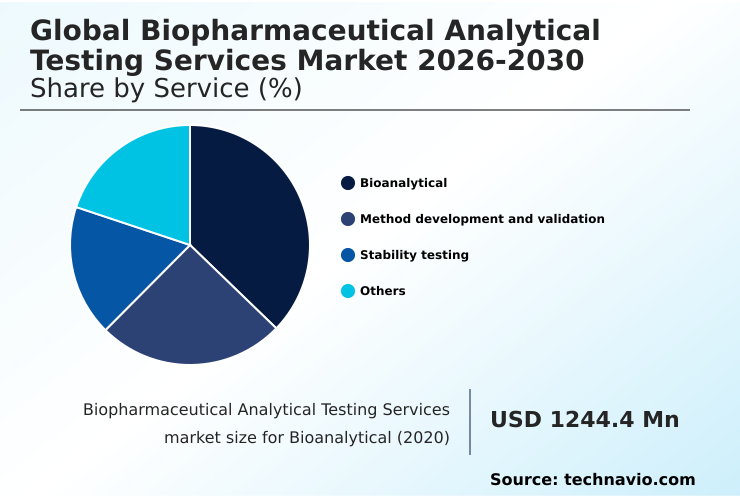
- By Service - Bioanalytical segment was valued at USD 2.03 billion in 2024
- By End-user - Pharma and biotech companies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Historic Market Opportunities 2020-2024: USD 9.01 billion
- Market Future Opportunities 2025-2030: USD 6.35 billion
- CAGR from 2025 to 2030 : 15.5%
Market Summary
- The biopharmaceutical analytical testing services market is expanding, with service providers now handling over 60% of all analytical testing for small to mid-sized biotech firms. This growth is propelled by the increasing complexity of biologic drugs, which require sophisticated characterization that many developers cannot support in-house, leading to a 15% year-over-year increase in outsourced stability testing programs.
- For instance, a typical operational scenario involves a startup outsourcing its entire method development and validation process to a specialized contract research organization (CRO). This strategic move mitigates the need for substantial capital investment in GMP-compliant laboratories and highly skilled personnel, which is a major market driver.
- However, the industry faces a significant challenge from an acute shortage of scientists with expertise in both advanced instrumentation and regulatory compliance, a factor that can create project bottlenecks and constrain the growth potential of service providers despite high demand.
What will be the Size of the Biopharmaceutical Analytical Testing Services Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Biopharmaceutical Analytical Testing Services Market Segmented?
The biopharmaceutical analytical testing services industry research report provides comprehensive data (region-wise segment analysis), with forecasts and analysis for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
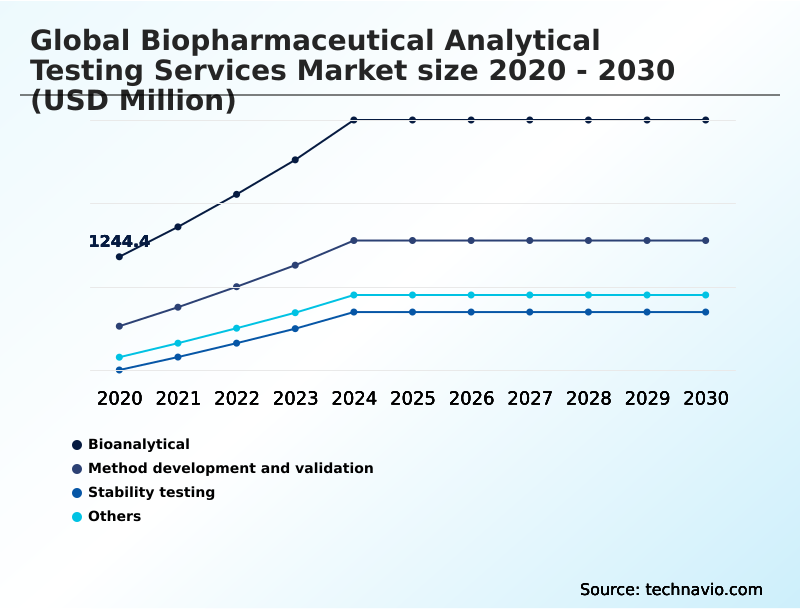
- Service
- Bioanalytical
- Method development and validation
- Stability testing
- Others
- End-user
- Pharma and biotech companies
- CROs
- Type
- Physiochemical analysis
- Biological assays
- Microbial testing
- Environmental monitoring
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- APAC
- China
- Japan
- India
- South America
- Brazil
- Argentina
- Middle East and Africa
- Saudi Arabia
- UAE
- South Africa
- Rest of World (ROW)
- North America
How is the Biopharmaceutical Analytical Testing Services Market Segmented by Service?
The bioanalytical segment is estimated to witness significant growth during the forecast period.
The market is segmented by service type, with bioanalytical solutions representing over 38% of revenue. This segment provides quantitative measurement of drugs using techniques like ligand-binding assays (LBA) for pharmacokinetics (PK) analysis, which improves trial success by over 15%.
Method development and validation focuses on creating robust procedures, including stability-indicating methods and applying analytical quality by design (aQbD) principles. Another critical segment is stability testing. Other services include essential lot release testing and raw material qualification.
This segmentation dictates operational focus; providers must offer everything from good laboratory practice (GLP) compliant bioanalysis and anti-drug antibody (ada) testing to GMP-certified sterility testing to meet diverse client needs across the development pipeline.
The Bioanalytical segment was valued at USD 2.03 billion in 2024 and showed a gradual increase during the forecast period.
How demand for the Biopharmaceutical Analytical Testing Services market is rising in the leading region?
North America is estimated to contribute 33.3% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Biopharmaceutical Analytical Testing Services Market demand is rising in North America Request Free Sample
The geographic landscape of the biopharmaceutical analytical testing services market is led by North America, which is projected to capture 33.3% of the market's incremental growth.
This dominance is sustained by the United States, which accounts for over 84% of the regional market and is home to the world's highest concentration of biopharmaceutical R&D activities.
In contrast, the APAC region is the fastest-growing market, with a CAGR of 16.0%, slightly outpacing North America's 15.8% CAGR.
This rapid expansion in APAC is driven by significant government investment in countries like China and South Korea, which are becoming major hubs for biosimilarity assessment and contract manufacturing.
The differing dynamics affect supply chains; North American providers focus on early-stage, complex physicochemical characterization, whereas APAC labs often scale for high-volume lot release testing and comparability studies, offering up to a 20% cost advantage for certain standardized assays.
What are the key Drivers, Trends, and Challenges in the Biopharmaceutical Analytical Testing Services Market?
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- Navigating the complexities of biopharmaceutical development requires specialized analytical support, particularly for next-generation therapeutics. Companies are increasingly seeking comprehensive gmp analytical services for cell therapy, which demand unique potency and characterization assays that differ significantly from traditional biologics.
- The use of mass spectrometry for monoclonal antibody characterization is now a cornerstone of development, providing deep insights into critical quality attributes that older techniques cannot resolve. This detailed analysis is vital for regulatory submissions and for establishing robust manufacturing processes.
- Many emerging companies find that outsourcing hplc method development and validation is a more efficient strategy, reducing internal capital expenditure by over 30% compared to building in-house capabilities. This approach also provides access to expertise in navigating the intricate ich guidelines for stability testing of biologics, ensuring that long-term data packages are compliant and reliable.
- As clinical programs advance, the reliance on specialized bioanalytical lab services for clinical trials becomes critical for generating the pharmacokinetic and immunogenicity data needed for regulatory approval, with top-tier labs demonstrating a 99% data acceptance rate by agencies.
- This strategic outsourcing model allows drug developers to focus on their core competencies while de-risking the complex analytical and regulatory pathway to market.
What are the key market drivers leading to the rise in the adoption of Biopharmaceutical Analytical Testing Services Industry?
- The increasing adoption of strategic outsourcing by pharmaceutical and biotechnology companies is a primary driver propelling market growth.
- The growing complexity of the biopharmaceutical pipeline is a primary market driver, with modern biologics requiring an arsenal of sophisticated techniques like high-performance liquid chromatography (hplc) and capillary electrophoresis for characterization.
- The rise of advanced therapies has created demand for entirely new analytical paradigms, such as those for adeno-associated virus (aav) analytics and mRNA vaccine analytics.
- Strategic outsourcing is another key driver, as it converts significant capital expenses into manageable operational costs, allowing a startup to save over 40% on initial lab setup.
- This model also provides immediate access to specialized expertise in areas like cell-based potency assays and host cell protein (hcp) analysis.
- The stringent regulatory landscape, demanding adherence to good manufacturing practices (gmp) and ICH Q6B guidelines, further compels developers to partner with compliant service providers.
What are the market trends shaping the Biopharmaceutical Analytical Testing Services Industry?
- A significant market trend is the intensifying focus on analytical services designed for advanced therapy medicinal products (ATMPs). This shift reflects the growing complexity of biopharmaceutical pipelines.
- A defining trend is the integration of advanced analytical technologies, with the multi-attribute method (MAM) improving efficiency by replacing up to five traditional QC tests with a single mass spectrometry (ms) run. This shift enables more comprehensive physicochemical characterization and post-translational modification (ptm) analysis on a routine basis.
- The adoption of process analytical technology (pat) is also growing, allowing for real-time monitoring and control, which reduces batch failure rates by 10%. This trend is driven by regulatory encouragement for deeper process understanding and the need to manage the complexity of novel biologics.
- Consequently, service providers are investing heavily in high-resolution instrumentation and data analytics platforms to offer more sophisticated services, including advanced CAR-T cell characterization and oligonucleotide analysis, moving beyond simple testing to become data interpretation partners.
What challenges does the Biopharmaceutical Analytical Testing Services Industry face during its growth?
- An acute shortage of skilled personnel, compounded by rising labor costs, presents a key challenge affecting industry growth.
- A formidable challenge is the shortage of skilled scientists, inflating labor costs by up to 20% in competitive biotech hubs and creating bottlenecks for complex tasks like method transfer validation. This scarcity impacts a provider's ability to perform a thorough immunogenicity risk assessment.
- The high capital investment in technology is another hurdle; rapid obsolescence means a cutting-edge instrument's value can diminish by over 50% in five years. This cycle pressures firms to constantly reinvest. Additionally, pricing pressure commoditizes routine services like bioburden testing and endotoxin testing (LAL).
- This forces providers to balance efficiency investments for standard assays against the high cost of innovation required for specialized services like extractables and leachables (e&l) testing and comparability studies, squeezing overall profit margins.
Exclusive Technavio Analysis on Customer Landscape
The biopharmaceutical analytical testing services market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the biopharmaceutical analytical testing services market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Biopharmaceutical Analytical Testing Services Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, biopharmaceutical analytical testing services market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Almac Group Ltd. - Specializing in biopharmaceutical analytics, services include comprehensive method development and validation for drug substances and products, supporting all phases of the development lifecycle.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Almac Group Ltd.
- Catalent Inc.
- Charles River Laboratories
- Element Materials Tech.
- Eurofins Scientific SE
- ICON plc
- Intertek Group Plc
- IQVIA Holdings Inc.
- Laboratory Corp.
- Lonza Group Ltd.
- Merck KGaA
- Parexel International Corp.
- Sartorius AG
- SGS SA
- Syneos Health
- Thermo Fisher Scientific Inc.
- WuXi AppTec Co. Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Market Intelligence Radar: High-Impact Developments & Growth Signals
- In the Life Sciences Tools and Services industry, the push for greater data integrity compliance, driven by stricter enforcement of 21 CFR Part 11, has increased the demand for validated analytical software and systems in the global biopharmaceutical analytical testing services market 2026-2030, impacting CMC regulatory submission strategies.
- The broader industry's adoption of real-time release testing (RTRT) methodologies is creating new opportunities for service providers that can offer advanced process analytical technology (PAT) and integrated data analytics, directly influencing approaches to in-process control (IPC) testing.
- A notable shift toward domestic biomanufacturing in key markets has intensified the need for local and responsive lot release testing and raw material qualification, altering supply chain dynamics for contract labs.
- The increasing investment in advanced therapeutic modalities like mRNA vaccine analytics and oligonucleotide analysis at the parent market level is mandating specialized analytical capabilities, such as adeno-associated virus (AAV) analytics, for biopharmaceutical testing partners.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Biopharmaceutical Analytical Testing Services Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 316 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 15.5% |
| Market growth 2026-2030 | USD 6347.3 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 13.6% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Argentina, Chile, Saudi Arabia, UAE, South Africa, Israel and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The biopharmaceutical analytical testing services ecosystem is a complex network where instrument manufacturers supply high-resolution mass spectrometers and chromatography systems, representing up to 40% of a laboratory's initial capital expenditure. Service providers, from global CROs to niche labs, leverage this technology to offer a range of services from early-stage physicochemical characterization to late-stage lot release testing.
- Their operations are strictly governed by regulatory bodies that mandate adherence to GMP and GLP standards, with audit failure rates below 2%. End-users, primarily pharma and biotech companies, outsource these services to accelerate development and mitigate risk. This interaction highlights a value chain where access to specialized technology and regulatory expertise is paramount.
- Supporting entities, including logistics providers specializing in cold-chain sample transport and software companies offering 21 CFR part 11 compliant data management systems, are integral to maintaining the integrity and efficiency of the entire process.
What are the Key Data Covered in this Biopharmaceutical Analytical Testing Services Market Research and Growth Report?
-
What is the expected growth of the Biopharmaceutical Analytical Testing Services Market between 2026 and 2030?
-
The Biopharmaceutical Analytical Testing Services Market is expected to grow by USD 6.35 billion during 2026-2030, registering a CAGR of 15.5%. Year-over-year growth in 2026 is estimated at 13.6%%. This acceleration is shaped by increasing strategic outsourcing by pharmaceutical and biotechnology companies, which is intensifying demand across multiple end-use verticals covered in the report.
-
-
What segmentation does the market report cover?
-
The report is segmented by Service (Bioanalytical, Method development and validation, Stability testing, and Others), End-user (Pharma and biotech companies, and CROs), Type (Physiochemical analysis, Biological assays, Microbial testing, and Environmental monitoring) and Geography (North America, Europe, APAC, South America, Middle East and Africa). Among these, the Bioanalytical segment is estimated to witness significant growth during the forecast period, driven by rising adoption across key application areas. Each segment includes detailed qualitative and quantitative analysis, along with historical data from 2020-2024 and forecasts through 2030 with year-over-year growth rates.
-
-
Which regions are analyzed in the report?
-
The report covers North America, Europe, APAC, South America and Middle East and Africa. North America is estimated to contribute 33.3% to market growth during the forecast period. Country-level analysis includes US, Canada, Mexico, Germany, UK, France, Italy, Spain, The Netherlands, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Argentina, Chile, Saudi Arabia, UAE, South Africa, Israel and Turkey, with dedicated market size tables and year-over-year growth for each.
-
-
What are the key growth drivers and market challenges?
-
The primary driver is increasing strategic outsourcing by pharmaceutical and biotechnology companies, which is accelerating investment and industry demand. The main challenge is acute shortage of skilled personnel and rising labor costs, creating operational barriers for key market participants. The report quantifies the impact of each driver and challenge across 2026 and 2030 with comparative analysis.
-
-
Who are the major players in the Biopharmaceutical Analytical Testing Services Market?
-
Key vendors include Almac Group Ltd., Catalent Inc., Charles River Laboratories, Element Materials Tech., Eurofins Scientific SE, ICON plc, Intertek Group Plc, IQVIA Holdings Inc., Laboratory Corp., Lonza Group Ltd., Merck KGaA, Parexel International Corp., Sartorius AG, SGS SA, Syneos Health, Thermo Fisher Scientific Inc. and WuXi AppTec Co. Ltd.. The report provides qualitative and quantitative analysis categorizing companies as dominant, leading, strong, tentative, and weak based on their market positioning. Company profiles include business segment analysis, SWOT assessment, key offerings, and recent strategic developments.
-
Market Research Insights
- The competitive landscape for biopharmaceutical analytical testing services is moderately fragmented, with the top ten providers accounting for less than 45% of the market share. This structure fosters a dynamic environment where leading contract research organizations continuously expand their capabilities.
- Recent strategic moves include acquisitions to enhance expertise in high-growth areas such as cell and gene therapy analytics, with some transactions valued at over 1.5 times the target's annual revenue. These investments are a direct response to the industry's demand for integrated service offerings that combine manufacturing support with comprehensive analytical packages.
- This trend towards strategic partnerships is reshaping client relationships. However, providers face intensifying pricing pressure for commoditized services, forcing them to focus on operational efficiency and differentiation through specialized, high-value testing for novel modalities.
We can help! Our analysts can customize this biopharmaceutical analytical testing services market research report to meet your requirements.


 RIA -
RIA - 
