Medical, Legal, And Regulatory (MLR) Review Software Market Size 2026-2030
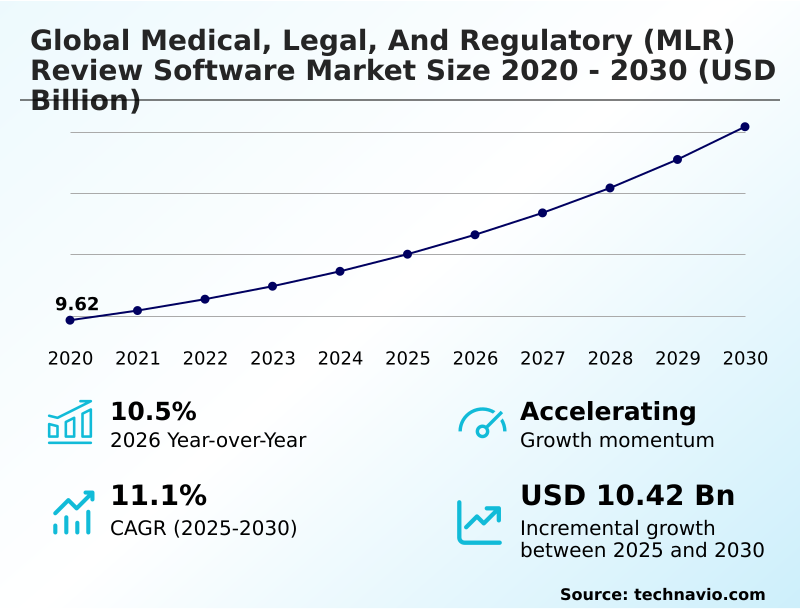
The Medical, Legal, And Regulatory (MLR) Review Software Market size was valued at USD 15.02 billion in 2025, growing at a CAGR of 11.1% during the forecast period 2026-2030.
Major Market Trends & Insights
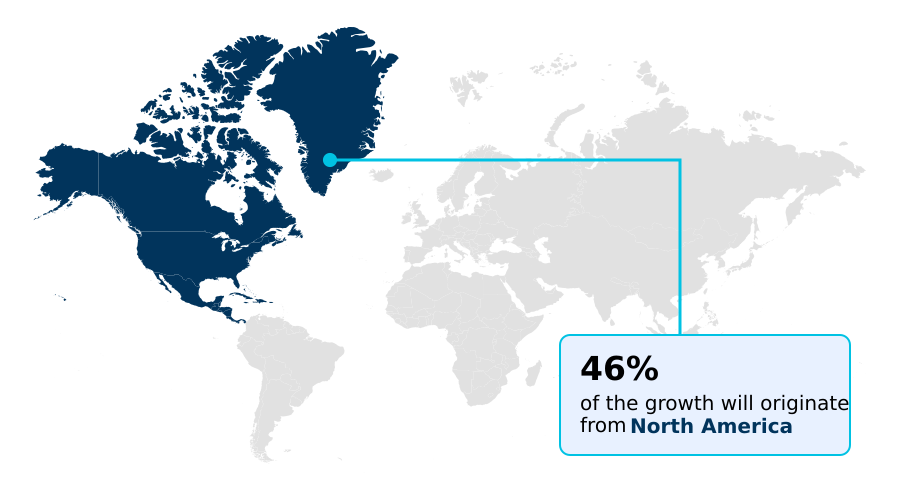
- North America dominated the market and accounted for a 45.9% growth during the forecast period.
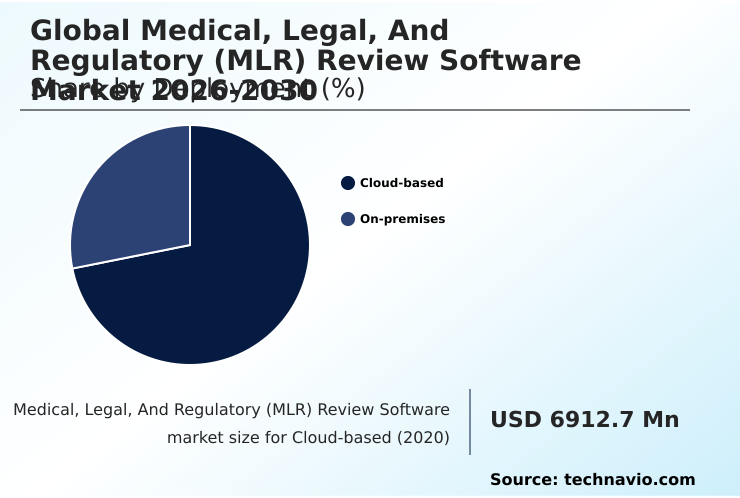
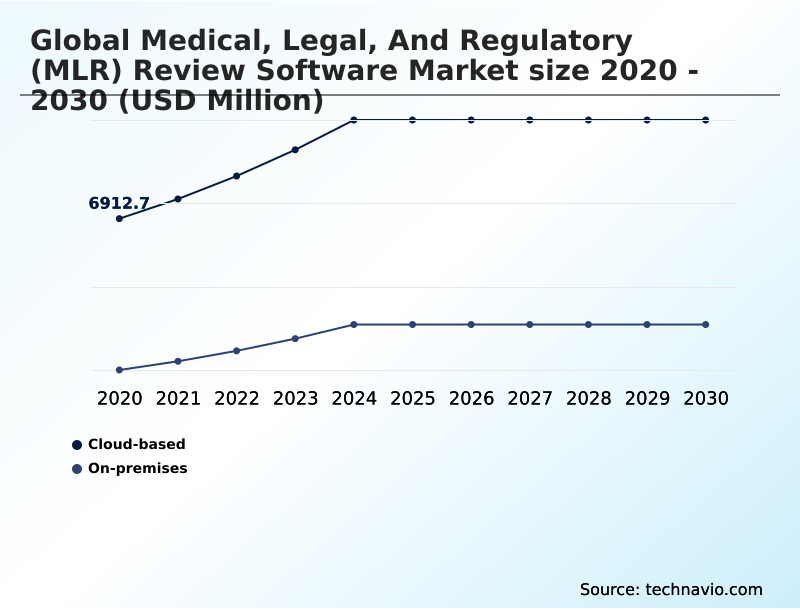
- By Deployment - Cloud-based segment was valued at USD 9.65 billion in 2024
- By Business Segment - Large enterprises segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Historic Market Opportunities 2020-2024: USD 15.83 billion
- Market Future Opportunities 2025-2030: USD 10.42 billion
- CAGR from 2025 to 2030 : 11.1%
Market Summary
- The medical, legal, and regulatory (MLR) review software market is defined by a strategic shift from manual, disconnected processes to centralized, automated platforms, which can reduce content approval cycle times by over 40%.
- A typical business scenario involves a pharmaceutical company launching a new product across 15+ countries, requiring a system that manages a global-to-local content model to ensure compliance with diverse health authority regulations. Adopting a unified platform improves claims substantiation accuracy by more than 95%, a critical factor for risk management.
- The market's momentum is propelled by the increasing complexity of global regulations and the proliferation of digital content for omnichannel engagement. However, growth is constrained by the significant financial and operational commitment required for implementation, including system validation and data migration, which poses a substantial barrier, particularly for smaller enterprises.
What will be the Size of the Medical, Legal, And Regulatory (MLR) Review Software Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Medical, Legal, And Regulatory (MLR) Review Software Market Segmented?
The medical, legal, and regulatory (mlr) review software industry research report provides comprehensive data (region-wise segment analysis), with forecasts and analysis for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
- Deployment
- Cloud-based
- On-premises
- Business segment
- Large enterprises
- SMEs
- End-user
- Pharmaceuticals
- Healthcare
- Biotechnology
- Food and beverage
- Geography
- North America
- US
- Canada
- Mexico
- APAC
- China
- India
- Japan
- Europe
- UK
- Germany
- France
- Middle East and Africa
- Saudi Arabia
- UAE
- Israel
- South America
- Brazil
- Argentina
- Rest of World (ROW)
- North America
How is the Medical, Legal, And Regulatory (MLR) Review Software Market Segmented by Deployment?
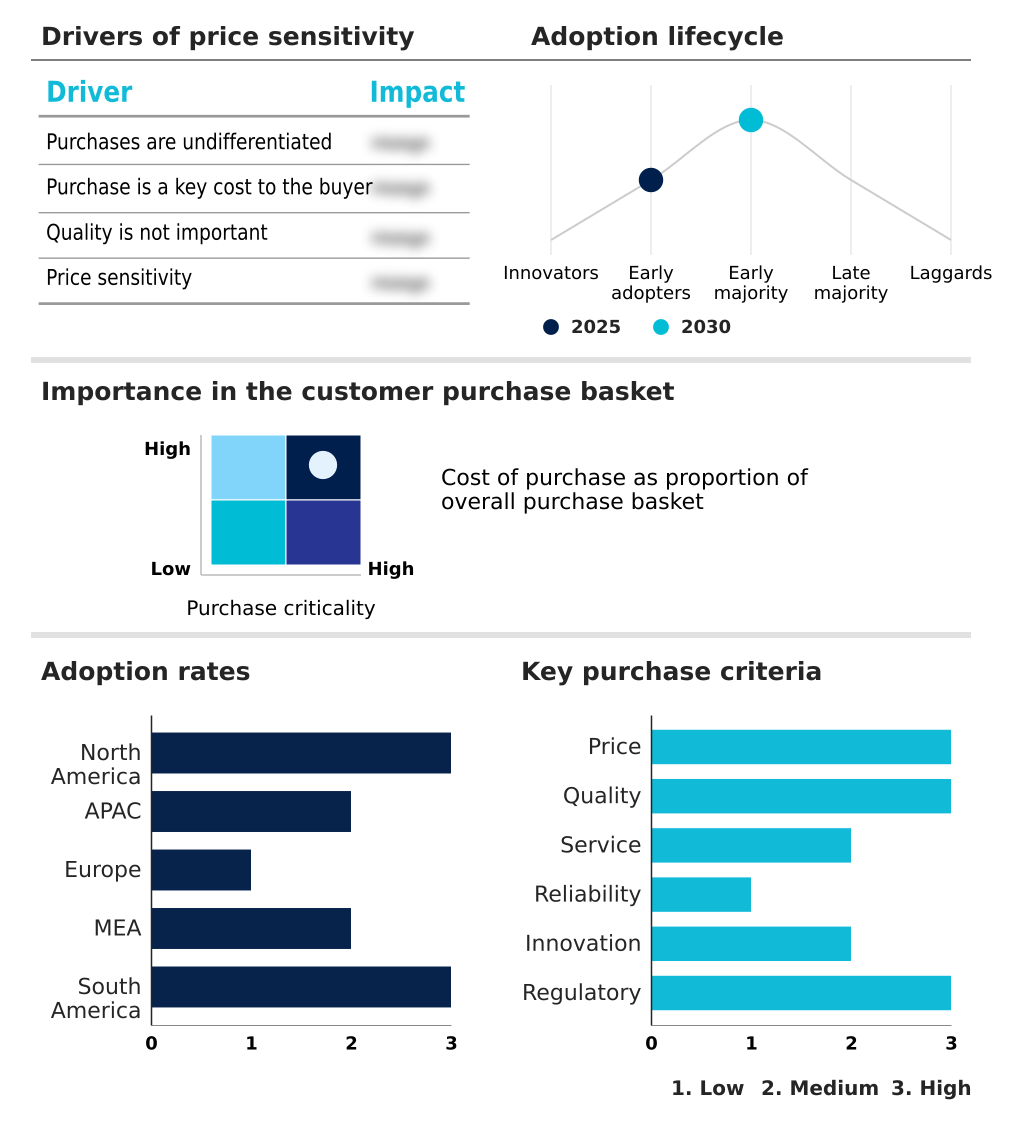
The cloud-based segment is estimated to witness significant growth during the forecast period.
Cloud-based deployment models represent over 70% of the medical, legal, and regulatory (MLR) review software market, driven by a compelling value proposition for life sciences firms.
This model, primarily delivered as software-as-a-service, shifts costs from capital to operational expenditures and can reduce initial infrastructure investment by more than 90% compared to on-premises solutions.
The core advantage lies in vendors managing the complex and continuous process of system validation and security, ensuring platforms adhere to standards like 21 CFR part 11 compliance. This outsourcing of maintenance frees up significant internal IT resources.
Furthermore, cloud-based delivery models offer superior accessibility, enabling seamless global-to-local content models and real-time collaboration that is essential for accelerating the content supply chain for promotional material review.
The Cloud-based segment was valued at USD 9.65 billion in 2024 and showed a gradual increase during the forecast period.
How demand for the Medical, Legal, And Regulatory (MLR) Review Software market is rising in the leading region?
North America is estimated to contribute 45.9% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Medical, Legal, And Regulatory (MLR) Review Software Market demand is rising in North America Request Free Sample
North America dominates the medical, legal, and regulatory (MLR) review software landscape, accounting for over 45% of the market, with the United States being the largest single market.
This leadership is a direct result of the region's stringent regulatory environment and highly litigious nature, making robust solutions for promotional material review a necessity.
In contrast, the APAC region is projected to exhibit the fastest growth at 11.8%, outpacing North America's 10.7% due to the expansion of local pharmaceutical industries and harmonization with global compliance standards.
European firms demonstrate a 15% higher adoption rate of unified platform approaches compared to emerging markets, a strategy used to manage disparate regulations across member states efficiently. This regional variance necessitates adaptable global-to-local content models and configurable automated workflow capabilities.
What are the key Drivers, Trends, and Challenges in the Medical, Legal, And Regulatory (MLR) Review Software Market?
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- For life sciences companies, navigating the promotional content landscape begins with a deep understanding of the mlr review process for pharma. The primary goal for commercial and medical affairs teams is to figure out how to streamline mlr approval, as delays directly impact revenue and competitive positioning.
- This investigation naturally leads to an evaluation of cloud-based mlr software benefits, which often include a 30-40% reduction in approval cycle times compared to legacy, on-premise systems.
- A key differentiator emerging among top solutions is the role of ai in mlr content review, where intelligent algorithms can pre-screen submissions and identify potential compliance risks, improving first-pass acceptance rates by over 25%.
- When selecting a platform, organizations weigh these technological advantages against the total cost of mlr review software, which extends beyond subscription fees to include implementation, validation, and training. The ultimate decision on the best mlr software for life sciences hinges on a platform’s ability to provide a validated, auditable, and efficient system that mitigates risk while accelerating speed-to-market.
- The best practices for mlr compliance are increasingly embedded within the software itself, guiding users toward creating compliant, effective communications from the outset.
What are the key market drivers leading to the rise in the adoption of Medical, Legal, And Regulatory (MLR) Review Software Industry?
- The market is primarily driven by increasing regulatory complexity and heightened enforcement scrutiny from global health authorities.
- The primary driver for the medical, legal, and regulatory (MLR) review software market is the dual pressure of increasing regulatory complexity and the exponential growth of digital content.
- A single global product launch can now require content adaptation for over 20 unique regulatory jurisdictions, a 30% increase in complexity compared to five years ago.
- This is compounded by the proliferation of content needed to support an omnichannel content strategy, with many firms seeing a 50% year-over-year rise in the volume of digital assets. This situation makes manual review processes inefficient and fraught with risk.
- Consequently, life sciences companies are compelled to adopt solutions with an automated workflow to ensure consistent compliance risk mitigation and maintain a defensible, immutable audit trail for all promotional material review activities.
What are the market trends shaping the Medical, Legal, And Regulatory (MLR) Review Software Industry?
- A transformative market trend is the integration of artificial intelligence and machine learning. This shift enables intelligent automation, moving beyond basic workflow management to predictive compliance.
- A dominant trend in the medical, legal, and regulatory (MLR) review software market is the deepening integration of artificial intelligence, which is evolving platforms from simple workflow automation tools to systems of intelligent automation.
- AI-powered pre-review features are becoming standard, capable of automatically identifying up to 70% of common compliance errors, such as missing fair balance information or unsubstantiated claims, before the content enters the formal review cycle. This is driven by the industry's need to reduce the workload on specialized reviewers and accelerate content velocity.
- The effect is a significant reduction in revision cycles and improved speed-to-market. Concurrently, the shift toward a unified platform approach enhances content lifecycle management, offering up to 25% greater visibility into global content reuse compared to fragmented, siloed systems.
What challenges does the Medical, Legal, And Regulatory (MLR) Review Software Industry face during its growth?
- A key challenge affecting market growth is the high total cost of ownership, compounded by the complexity of platform implementation and validation.
- A formidable challenge constraining the medical, legal, and regulatory (MLR) review software market is the high total cost of ownership, where initial license fees can account for less than 40% of the first-year investment.
- This is caused by substantial ancillary costs associated with system validation, data migration from legacy systems, and integration with other enterprise platforms, a process that can take over six months to complete. The effect is a significant barrier to entry for small and medium-sized enterprises, which often lack the necessary capital and specialized IT resources.
- Furthermore, the complexity of implementation can lead to organizational resistance, with user adoption rates in some departments falling 25% below initial targets without a robust change management strategy to drive process harmonization.
Exclusive Technavio Analysis on Customer Landscape
The medical, legal, and regulatory (mlr) review software market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the medical, legal, and regulatory (mlr) review software market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Medical, Legal, And Regulatory (MLR) Review Software Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, medical, legal, and regulatory (mlr) review software market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Acheron Software Consultancy - Key solutions encompass unified, cloud-based platforms that automate and document the content review lifecycle, ensuring regulatory adherence and accelerating market access for life sciences organizations.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Acheron Software Consultancy
- Adobe Inc.
- Anthill
- Aprimo
- Cegedim SA.
- Compliance Path
- Framework Solutions LLC
- Generis Enterprise
- IQVIA Holdings Inc.
- MasterControl Solutions Inc.
- Open Text Corp.
- Optum Inc.
- Red Marker
- Salesforce Inc.
- Trinity Consultants
- Veeva Systems Inc.
- Vodori
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Market Intelligence Radar: High-Impact Developments & Growth Signals
- In the application software industry, the pervasive integration of artificial intelligence and machine learning into enterprise platforms is creating a new standard for operational efficiency. This directly impacts the medical, legal, and regulatory (MLR) review software market by driving demand for intelligent automation features, such as predictive compliance checks and automated claims substantiation, to enhance compliance risk mitigation.
- The widespread enterprise shift toward subscription-based, cloud-native software-as-a-service (SaaS) models is fundamentally altering procurement and IT cost structures. This accelerates adoption in the medical, legal, and regulatory (MLR) review software market, where cloud-based delivery models lower upfront investment and simplify validation, making advanced compliance tools more accessible.
- Heightened global focus on data privacy and security regulations, such as GDPR and CCPA, is forcing application software vendors to embed robust governance and security-by-design principles into their products. For the medical, legal, and regulatory (MLR) review software market, this translates into a critical need for platforms that guarantee data residency, encryption, and an immutable audit trail to protect sensitive health information.
- The move toward highly interconnected and interoperable enterprise application ecosystems, where CRM, ERP, and DAM systems communicate seamlessly, is reshaping workflow expectations. This development pressures the medical, legal, and regulatory (MLR) review software market to provide robust APIs and pre-built connectors that support a unified content supply chain and enable a cohesive omnichannel content strategy.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Medical, Legal, And Regulatory (MLR) Review Software Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 289 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 11.1% |
| Market growth 2026-2030 | USD 10423.0 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 10.5% |
| Key countries | US, Canada, Mexico, China, India, Japan, Australia, South Korea, Indonesia, UK, Germany, France, Italy, Spain, The Netherlands, Saudi Arabia, UAE, Israel, South Africa, Turkey, Brazil, Argentina and Chile |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The medical, legal, and regulatory (MLR) review software market ecosystem is a specialized value chain designed to ensure life sciences content compliance. The ecosystem begins with technology providers supplying cloud infrastructure and AI algorithms, which software vendors leverage to build their platforms.
- These vendors, which constitute the core of the market, must ensure their solutions facilitate 21 CFR Part 11 compliance to meet the stringent requirements of regulatory bodies like the FDA. End-users—primarily pharmaceutical and biotech firms, with large enterprises representing over 60% of the customer base—are the primary drivers of demand.
- The interaction is centered on risk mitigation and efficiency; a validated MLR platform can increase reviewer capacity by more than 20% while providing an immutable audit trail essential for defending against regulatory actions.
What are the Key Data Covered in this Medical, Legal, And Regulatory (MLR) Review Software Market Research and Growth Report?
-
What is the expected growth of the Medical, Legal, And Regulatory (MLR) Review Software Market between 2026 and 2030?
-
The Medical, Legal, And Regulatory (MLR) Review Software Market is expected to grow by USD 10.42 billion during 2026-2030, registering a CAGR of 11.1%. Year-over-year growth in 2026 is estimated at 10.5%%. This acceleration is shaped by increasing regulatory complexity and heightened enforcement scrutiny, which is intensifying demand across multiple end-use verticals covered in the report.
-
-
What segmentation does the market report cover?
-
The report is segmented by Deployment (Cloud-based, and On-premises), Business Segment (Large enterprises, and SMEs), End-user (Pharmaceuticals, Healthcare, Biotechnology, and Food and beverage) and Geography (North America, APAC, Europe, Middle East and Africa, South America). Among these, the Cloud-based segment is estimated to witness significant growth during the forecast period, driven by rising adoption across key application areas. Each segment includes detailed qualitative and quantitative analysis, along with historical data from 2020-2024 and forecasts through 2030 with year-over-year growth rates.
-
-
Which regions are analyzed in the report?
-
The report covers North America, APAC, Europe, Middle East and Africa and South America. North America is estimated to contribute 45.9% to market growth during the forecast period. Country-level analysis includes US, Canada, Mexico, China, India, Japan, Australia, South Korea, Indonesia, UK, Germany, France, Italy, Spain, The Netherlands, Saudi Arabia, UAE, Israel, South Africa, Turkey, Brazil, Argentina and Chile, with dedicated market size tables and year-over-year growth for each.
-
-
What are the key growth drivers and market challenges?
-
The primary driver is increasing regulatory complexity and heightened enforcement scrutiny, which is accelerating investment and industry demand. The main challenge is high total cost of ownership and implementation complexity, creating operational barriers for key market participants. The report quantifies the impact of each driver and challenge across 2026 and 2030 with comparative analysis.
-
-
Who are the major players in the Medical, Legal, And Regulatory (MLR) Review Software Market?
-
Key vendors include Acheron Software Consultancy, Adobe Inc., Anthill, Aprimo, Cegedim SA., Compliance Path, Framework Solutions LLC, Generis Enterprise, IQVIA Holdings Inc., MasterControl Solutions Inc., Open Text Corp., Optum Inc., Red Marker, Salesforce Inc., Trinity Consultants, Veeva Systems Inc. and Vodori. The report provides qualitative and quantitative analysis categorizing companies as dominant, leading, strong, tentative, and weak based on their market positioning. Company profiles include business segment analysis, SWOT assessment, key offerings, and recent strategic developments.
-
Market Research Insights
- The competitive landscape for medical, legal, and regulatory (MLR) review software is highly concentrated, with the top three vendors commanding over 60% of the market. Leading providers like Veeva Systems and Adobe are differentiating through innovation in artificial intelligence, launching features such as AI-powered pre-review that can reduce initial reviewer workload by up to 30%.
- This focus on intelligent automation directly addresses the life sciences industry's need for greater efficiency and accelerated speed-to-market for promotional materials. These developments are creating a technology race centered on enhancing workflow interoperability and providing robust predictive analytics.
- However, vendors must navigate the persistent challenge of ensuring data privacy and continuous validation in the cloud to meet stringent regulatory standards like 21 CFR Part 11.
We can help! Our analysts can customize this medical, legal, and regulatory (mlr) review software market research report to meet your requirements.


 RIA -
RIA - 
