Medical Writing Market Size 2024-2028
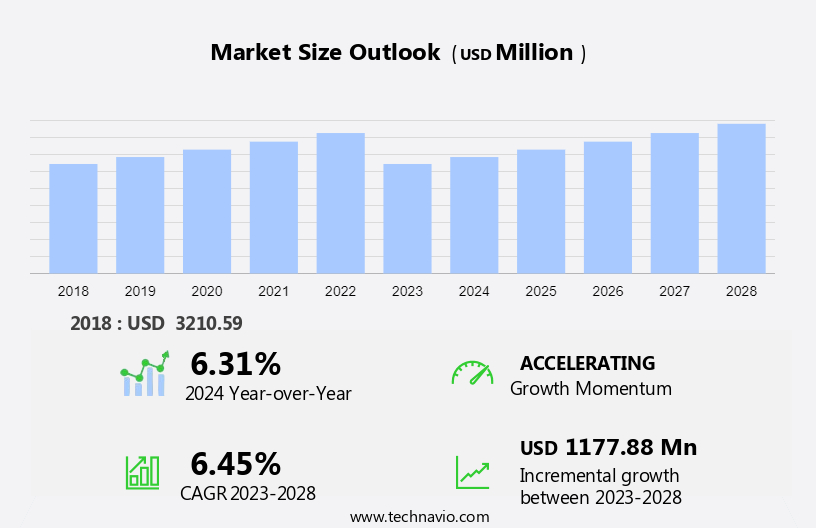
The medical writing market size is forecast to increase by USD 1.18 billion, at a CAGR of 6.45% between 2023 and 2028.
- The market growth depends on key drivers such as the increase in the number of clinical trials. The medical writing market plays a crucial role in scientific data analysis, regulatory submissions, and the creation of educational materials. As the healthcare industry invests heavily in evidence-based medicine, skilled medical writers are in demand to communicate complex scientific information effectively. A significant trend shaping the market is the increasing adoption of AI in medical writing, which enhances efficiency and accuracy in document creation. However, a key challenge affecting the market growth is data security and privacy concerns associated with medical writing, especially when handling sensitive patient and clinical trial information.
What will be the Size of the Market During the Forecast Period?
- The market encompasses various sectors, including patient information leaflets, scientific manuscripts, educational materials, regulatory writing, clinical writing, and medical writing sessions. These materials are essential for physicians and healthcare professionals to effectively communicate complex medical information to patients and peers. The market is significantly influenced by advancements in genetic engineering and bioinformatics, which require precise and accurate documentation. Clinical data management is another critical area that relies on medical writing for the collection, analysis, and reporting of clinical trial data. The market for medical writing continues to grow as the demand for clear and concise communication in the medical field increases.
How is this market segmented and which is the largest segment?
The market research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD billion" for the period 2024-2028, as well as historical data from 2018-2022 for the following segments.
- Type
- Clinical writing
- Regulatory writing
- Others
- End-user
- Pharmaceutical
- biotech companies
- Contract research organization
- others
- Geography
- North America
- US
- Asia
- China
- India
- Europe
- Germany
- UK
- Rest of World (ROW)
- North America
By Type Insights
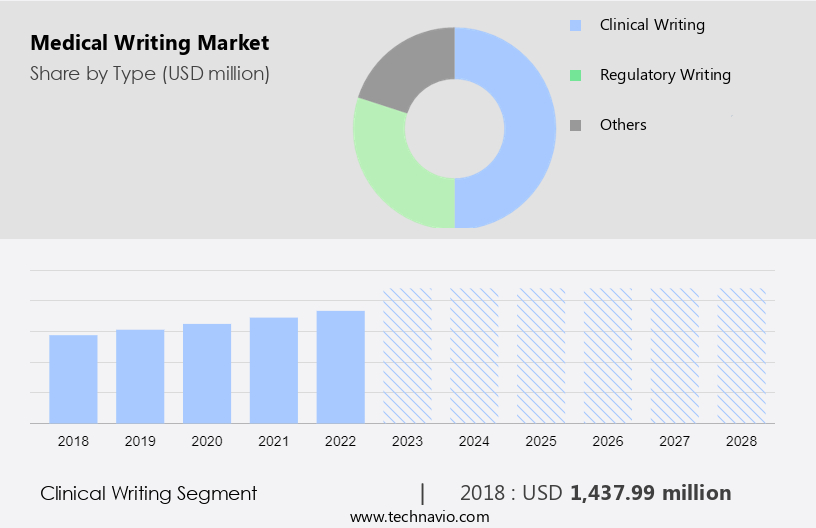
- The clinical writing segment is estimated to witness significant growth during the forecast period.
Clinical writing refers to the type of writing that healthcare professionals engage in regularly. Examples of clinical writing include documenting progress or treatment notes in medical records, updating patient charts, preparing referral and consultation letters, and completing various administrative forms. This form of writing communicates essential, accurate, and detailed information regarding a patient's condition, diagnostic tests, treatment plans, and prognosis. Unlike other forms of medical writing, clinical writing directly affects patient care. Additionally, it carries legal implications and may be used as evidence in malpractice or negligence lawsuits.
Get a glance at the market report of share of various segments Request Free Sample
The clinical writing segment was valued at USD 1.48 billion in 2018 and showed a gradual increase during the forecast period.
Regional Analysis
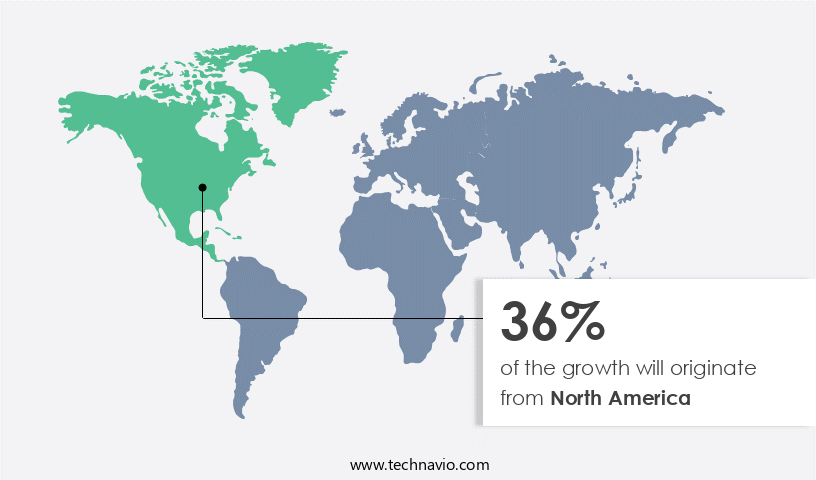
- North America is estimated to contribute 36% to the growth of the global market during the forecast period.
Technavio's analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
For more insights on the market share of various regions Request Free Sample
The market is thriving due to the region's emphasis on evidence-based medicine and the substantial healthcare expenditure. With the increasing prevalence of diseases worldwide, there is a growing demand for high-quality scientific data and patient information leaflets. This need is met through the production of scientific manuscripts, educational materials, and regulatory submissions. Skilled medical writers play a crucial role in transforming complex scientific research into clear and concise language for various audiences, including physicians, patients, and regulatory bodies. The market encompasses a wide range of applications, including research articles, conference papers, and documentation for drug-related information, medical device regulations, and study protocols.
Moreover, advancements in medical technologies, such as genetic engineering, bioinformatics, and agriculture biotechnology, necessitate the need for comprehensive clinical data management and medical writing sessions. The internship forum provides opportunities for aspiring medical writers to gain valuable experience and contribute to the development of medication innovations and medical apparatus regulations. The internet has revolutionized the medical writing landscape, enabling the dissemination of medical journalism and real-time access to drug, device, biologic, and autologous molecule information. The market is expected to continue growing as the need for clear and accurate communication in the healthcare sector persists.
Market Dynamics
Our researchers analyzed the data with 2023 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
What are the key market drivers leading to the rise in adoption of Medical Writing Market?
Increase in number of clinical trials is the key driver of the market.
- The market encompasses various sectors, including scientific data analysis, creation of patient information leaflets, and the crafting of scientific manuscripts for medical journals and educational materials. This field plays a pivotal role in medical communication, catering to the needs of physicians, patients, and regulatory bodies. Scientific data is meticulously analyzed and presented in a clear and concise manner through clinical writing, research articles, conference papers, and regulatory submissions. The healthcare industry's substantial expenditure on evidence-based medicine necessitates the involvement of skilled medical writers to ensure accurate and effective communication of complex scientific information. Medical writing extends to medical education, where it is essential to convey intricate concepts to students and professionals.
- Regulatory submissions for autologous molecules, drugs, devices, biologics, and medical apparatus require precise documentation to meet regulatory requirements. The advent of technologies such as genetic engineering, bioinformatics, and agriculture biotechnology has expanded the scope of medical writing. Clinical data management, medical writing sessions, internship forums, and regulatory paperwork are integral components of this dynamic field. Medical writing also encompasses medical journalism, where accurate and unbiased reporting on medication innovations, clinical trials, and drug-related information is crucial. The internet has revolutionized the dissemination of medical information, further expanding the role of medical writers in the digital age.
What are the market trends shaping the Medical Writing Market?
The increasing adoption of AI in medical writing is the upcoming trend in the market.
- The market encompasses various sectors, including scientific data analysis, creation of patient information leaflets, and the crafting of scientific manuscripts for medical journals and educational materials. This field plays a pivotal role in medical communication, catering to the needs of physicians, patients, and regulatory bodies. Scientific data is a crucial component of medical writing, as it forms the basis for evidence-based medicine and clinical research. Medical writers are responsible for interpreting complex scientific data and translating it into clear, concise, and accurate documentation. Patient information leaflets are another essential aspect of medical writing, as they provide vital information to patients about their medications, treatments, and medical devices.
- Medical writers ensure that these leaflets are easy to understand, informative, and comply with regulatory requirements. Scientific manuscripts, research articles, and conference papers are also integral parts of the market. They serve to disseminate new medical knowledge, innovations, and research findings to the global medical community. The healthcare industry's increasing expenditure on research and development has led to a growing demand for skilled medical writers. They are required to produce regulatory submissions, study protocols, and documentation for clinical trials, medical apparatus regulations, and drug-related information. The advent of technologies such as genetic engineering, bioinformatics, and agriculture biotechnology has further expanded the scope of medical writing.
- Clinical data management and medical writing sessions are essential components of these advanced research areas. Internship forums provide opportunities for aspiring medical writers to gain practical experience and learn from industry experts. Regulatory paperwork and documentation are also essential aspects of the market, ensuring that drugs, devices, biologics, and other medical innovations meet regulatory requirements.
What challenges does Medical Writing Market face during the growth?
Data security and privacy concerns associated with medical writing is a key challenge affecting the market growth.
- The market encompasses various sectors, including scientific data analysis, creation of patient information leaflets, and the crafting of scientific manuscripts for medical journals and educational materials. This field plays a pivotal role in medical communication, catering to the needs of physicians, patients, and regulatory bodies. Scientific data is meticulously analyzed and presented in a clear and concise manner through clinical writing, research articles, conference papers, and regulatory submissions. The healthcare industry's substantial expenditure on evidence-based medicine necessitates the involvement of skilled medical writers to ensure accurate and effective communication of complex scientific information. Medical writing extends to medical education, where it is essential to convey intricate concepts to students and professionals.
- Regulatory submissions for autologous molecules, drugs, devices, biologics, and medical apparatus require precise documentation to meet regulatory requirements. The advent of technologies such as genetic engineering, bioinformatics, and agriculture biotechnology has expanded the scope of medical writing. Clinical data management, medical writing sessions, internship forums, and regulatory paperwork are integral components of this dynamic field. Medical writing also encompasses medical journalism, where accurate and unbiased reporting on medication innovations, clinical trials, and drug-related information is crucial. The internet has revolutionized the dissemination of medical information, further expanding the role of medical writers in the digital age.
Exclusive Customer Landscape
The market forecasting report includes the adoption lifecycle of the market, covering from the innovator's stage to the laggard's stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape
Key Companies & Market Insights
Companies are implementing various strategies, such as strategic alliances, market forecast , partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the market. The market research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Cactus Communication
- Cardinal Health Inc.
- Certara Inc.
- Clinovo
- Freyr Software Services
- ICON plc
- InClin Inc.
- Indegene Pvt. Ltd.
- IQVIA Holdings Inc.
- MakroCare
- OMICS International Pvt. Ltd.
- Quanticate International Ltd.
- SIRO Clinpharm Pvt. Ltd.
- Syneos Health Inc.
- Trilogy Writing and Consulting GmbH
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key market players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Research Analyst Overview
In the dynamic landscape of healthcare communications, medical writing plays a pivotal role in shaping the narrative around scientific discoveries and clinical advancements. The market encompasses a broad spectrum of services, including regulatory writing, clinical trial reports, medical education materials, and scientific publications. Patients, healthcare providers, and regulatory bodies rely on accurate, clear, and concise medical communications to make informed decisions. As a result, the demand for skilled medical writers continues to increase. The market for medical writing services is driven by several factors, including the growing number of clinical trials, the increasing complexity of regulatory requirements, and the rising importance of medical education in healthcare.
Further, infusing scientific accuracy and effective communication, medical writing is a crucial component of the healthcare ecosystem. Its influence extends to various sectors, including pharmaceuticals, biotechnology, and medical devices. With the continuous evolution of healthcare and the increasing emphasis on evidence-based medicine, the role of medical writing in shaping the healthcare narrative is more significant than ever. The market for medical writing services is expected to grow significantly in the coming years, driven by the increasing demand for clear and concise medical communications. The use of advanced technologies, such as artificial intelligence and machine learning, is also expected to transform the medical writing landscape, making it more efficient and effective.
|
Market Scope |
|
|
Report Coverage |
Details |
|
Page number |
146 |
|
Base year |
2023 |
|
Historic period |
2018-2022 |
|
Forecast period |
2024-2028 |
|
Growth momentum & CAGR |
Accelerate at a CAGR of 6.45% |
|
Market Growth 2024-2028 |
USD 1.18 billion |
|
Market structure |
Fragmented |
|
YoY growth 2023-2024(%) |
6.31 |
|
Key countries |
US, Germany, UK, China, and India |
|
Competitive landscape |
Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
What are the Key Data Covered in this Market Research and Growth Report?
- CAGR of the market during the forecast period
- Detailed information on factors that will drive the market growth and forecasting between 2024 and 2028
- Precise estimation of the size of the market and its contribution of the market in focus to the parent market
- Accurate predictions about upcoming market growth and trends and changes in consumer behaviour
- Growth of the market across North America, Asia, Europe, and Rest of World (ROW)
- Thorough analysis of the market's competitive landscape and detailed information about companies
- Comprehensive analysis of factors that will challenge the growth of market companies
We can help! Our analysts can customize this market research report to meet your requirements.


 RIA -
RIA - 
